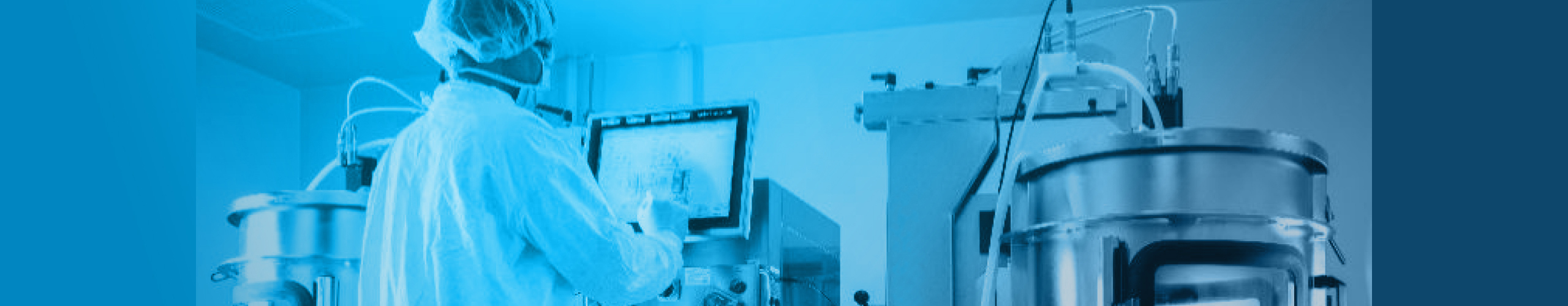
ADHERENT PROCESSES
ABL has extensive experience utilizing cell factories and hyperstacks
We can scale-up your process up to 60 sqm (24xCF40).
We have performed a significant number of batches with Vaccinia and MVA (Modified Ankara Virus) and other Pox viruses, hence we have established a procedure to perform Aseptic Process Simulation (APS) which enables us to accelerate the release of your first clinical batch with overall cost reduction achievement.
Dowstream Process (DSP) has been optimized using specific equipment to reduce manufacturing cycle time and cost with the highest level of quality.
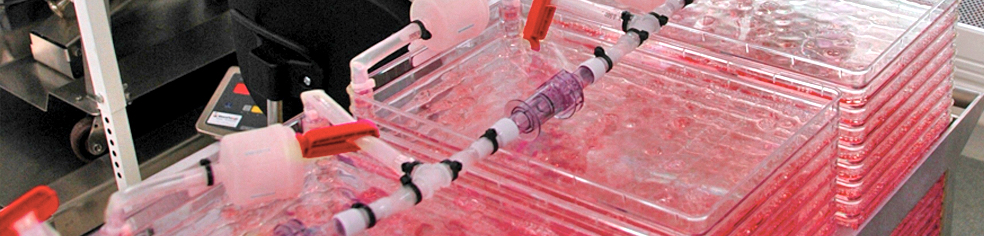
Cell culture on microcarriers
Scalability of your adherent cell process might face technical challenges to address the needs of your therapeutic indication – We offer to overcome the scalability related challenges developing the USP using Microccarriers in Single Use Bioreactors
Adherent platforms remain the most widely used technologies to produce specific types of virus. At ABL we have scaled-up many projects in Cell Factory. However depending on the therapy you want to address (dosage, number of injections per patient, number of patients) we may face technical limitations and challenges to fix and control several key parameters of the USP (upstream process).
Adapting an adherent process to suspension could positively impact the yield and quality attributes of your compound.
At ABL, we can mitigate the risk using Microcarriers allowing growth of adherent cells in suspension. It has resulted in significant cell culture surfaces compared to the same processes performed in Cell Factory, in a 200L single use bioreactor we have achieved >120 sqm, which is equivalent to 48xCF40.
ABL has already successfully developed processes with microcarriers in single use bioreactor. We apply a methodology consisting in screening commercial microcarriers supported by media and buffers optimization through a DOE approach.
Roller Bottle
We are equipped with a RC40 roller bottle resulting in a surface of culture up to 16 sqm.
At the Strasbourg and Rockville sites, ABL is equipped with automated systems to enable a process scale-up.
Single use fixed-bed bioreactors platform
We are equipped with laboratory and up to industrial scale fixed-bed bioreactors to bolster your program with adherent cells which cannot be cultivated in single-use bioreactor (pure suspended or using microcarriers).
The market you want to address requires large quantities of viral vectors, the fixed-bed bioreactor technology represents a manufacturing cycle time and cost-efficient alternative to multiple CF40 process.
Our laboratory equipment is a reliable scale-down model to perform small scale production and to support process development which can easily be transferred to the manufacturing equipment.